How far can LiIon be discharged?

I do often see a question about how far LiIon can be discharged and if it is safe to charge a discharged LiIon. These questions can be fairly difficult to answer precisely, LiIon manufactures do not publish much about it and LiIon is not just LiIon, there are many different chemistries with similar but not equal specifications.
How to measure the discharge cut-off voltage?
Some people do not wish to discharge below 3.6 volt (or something), but why stop there when the specifications say 3.0 or lower?
There is some sort of reason for this and it has to do with how you measure voltage: Usual voltage is measured with the battery out of the equipment, i.e. the battery is unloaded. Here is an example:
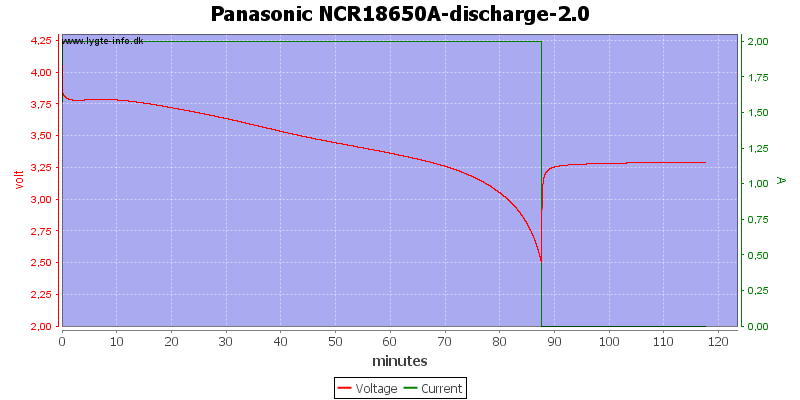
Here I discharge a cell to 2.5 volt with 2A, when I stop the voltage jumps to 3.3 volt and it jumps fairly fast.
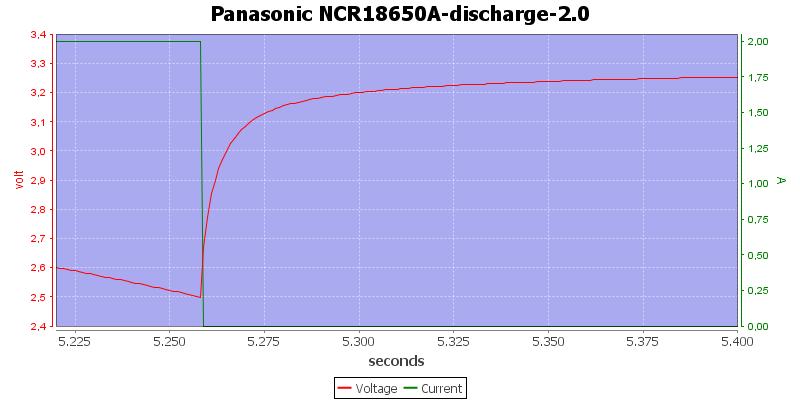
Zooming in on the stop, the curve shows 3 minutes. It takes 12 seconds for the voltage to raise from 2.5 volt to 3.1 volt. This means that if you take the battery out and measures it you will never see the 2.5 volt.
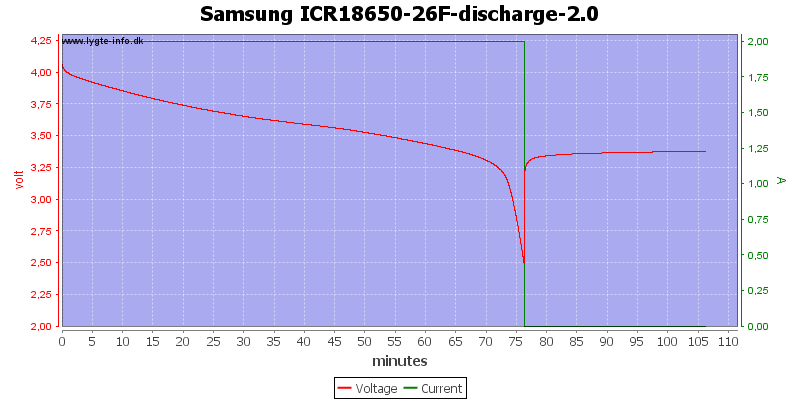
Lets try with another cell.
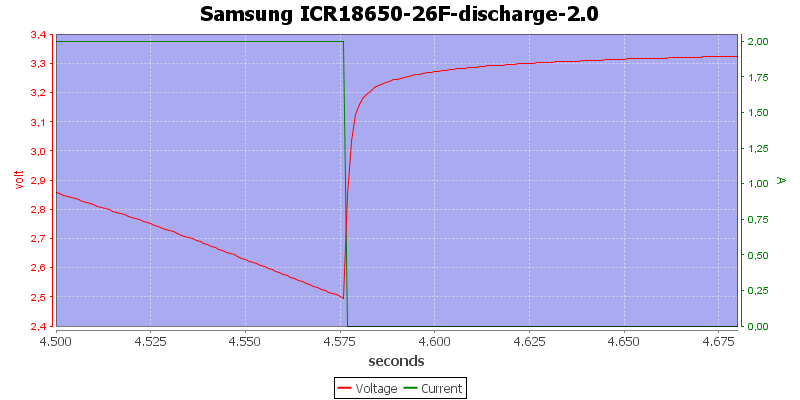
This is also a 3 minute view, the voltage raises from 2.5 volt to 3.1 volt in 3 seconds and in 7 seconds it is about 3.2 volt. Again it is not possible to measure the voltage where the equipment turns off.
How far the voltage jumps depends on the discharge current, a lower current will give a smaller jump and on the battery chemistry, different chemistries gives different voltage jumps. This means that measuring voltage with the battery out of the equipment will not give a correct indication of the minimum voltage the equipment discharged the battery to.
Due to the above jump in voltage it is rather difficult to know what the actual battery voltage is inside the flashlight when it is on and the safe way is to assume it is empty when the unloaded voltage is below some value. The 3.6 volt is fine for some batteries, but it is possible to go lower with many modern cells as seen above. The table at the end of the remaning capacity article can also be used.
How far to discharge batteries during normal use?
The first answer to this is easy, check the datasheet:

Here are some values from 4 different datasheets. The most common values are 2.75V and 2.5V today.
These values are the lowest voltage that is allowed on the battery if the full lifetime must be reached*. This means that for any normal usage this is the lowest value to discharge the battery to. This voltage do not include any current, i.e. the limit is not one that can only be discharged to at high currents, it is allowed to discharge with a very low current to this specification.
It is not within specifications to discharge below this voltage, just because the battery will recover to above this voltage when the load is removed.
*Usual, the 2.0 volt specification is not used for lifetime tests on that cell, there the lower limit is 2.5 volt.
But my over-discharge protection do first trip at 2.3 volt, is it faulty?
Usual no, protection is normally not used, i.e. it is allowed to go outside the range for best lifetime.

Here is some data from Samsung, the over-discharge protection is supposed to be outside the normal usage range.
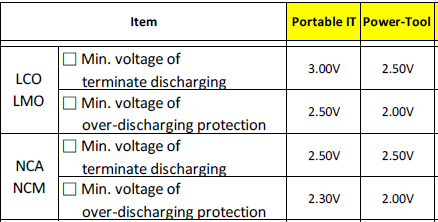
And if I include a bit more of the table it can be seen that it varies with application. At high current the protection can be at a lower voltage.
From this table it looks like it is acceptable to discharge LCO/LMO (Real ICR/IMR) cells below minimum value, when it is done at high current, but with the more modern cells it is best not discharging too much below rated voltage.
How low voltage is safe to recharge?
Here are some illustrations from LiIon handbooks:
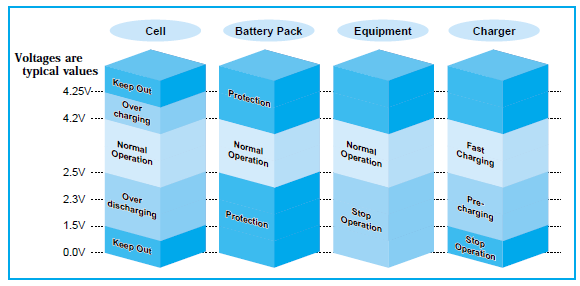
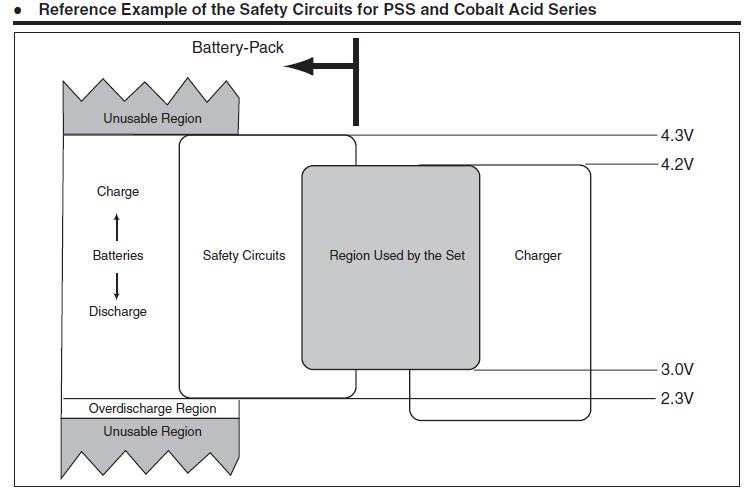
In both illustrations there is:
Operating range, that is the voltage range from the datasheet.
Protection/safety range, that covers a wider voltage range, but may add extra wear to the battery.
Keep out/unusable range, that is very low or very high voltage and the battery is dangerous.

The Samsung guidelines also list a minimum voltage.
These 3 items gives the minimum voltage as 1.5 volt, somewhere below 2.3 volt and 1 volt. This covers different brands and different chemistries.
Another importance detail is pre-charging, when the voltage is low the battery must not be charged at full current. A reduced current must be used.
When a battery is discharged to this low voltage it may lose some capacity and will lose some lifetime.
The reason to not charge very discharged batteries is because the chemistry breaks down at low voltages, depending on how much the chemistry has degraded the battery may explode when charged!
Conclusion
With the above as basis I will recommend the following:
1) Do not use over discharge protection as signal for when to charge batteries, it might wear the batteries down faster.
2) Do not charge batteries measuring below 2 volt, except if you have data saying it is safe.
3) When cells are down to 3.3 to 3.6 volt unloaded they are empty according to my tests, when the load is a few ampere.
4) Just because the cell recovers voltage, do not make it advisable to discharge below the limits while current is on.
And remember that if a battery gets hotter than usual during charger and/or will not charge fully (i.e. charger will not terminate), it is dead and maybe dangerous.
Notes
This article uses excerpts from:
Lithium Ion Rechargeable Batteries Technical Handbook (Sony)
Lithium Ion Batteries Technical Handbook (Panasonic)
Pack Design Guideline (Samsung datasheet, I have done some cut and paste in the table)
More voltage jumps with and without load










